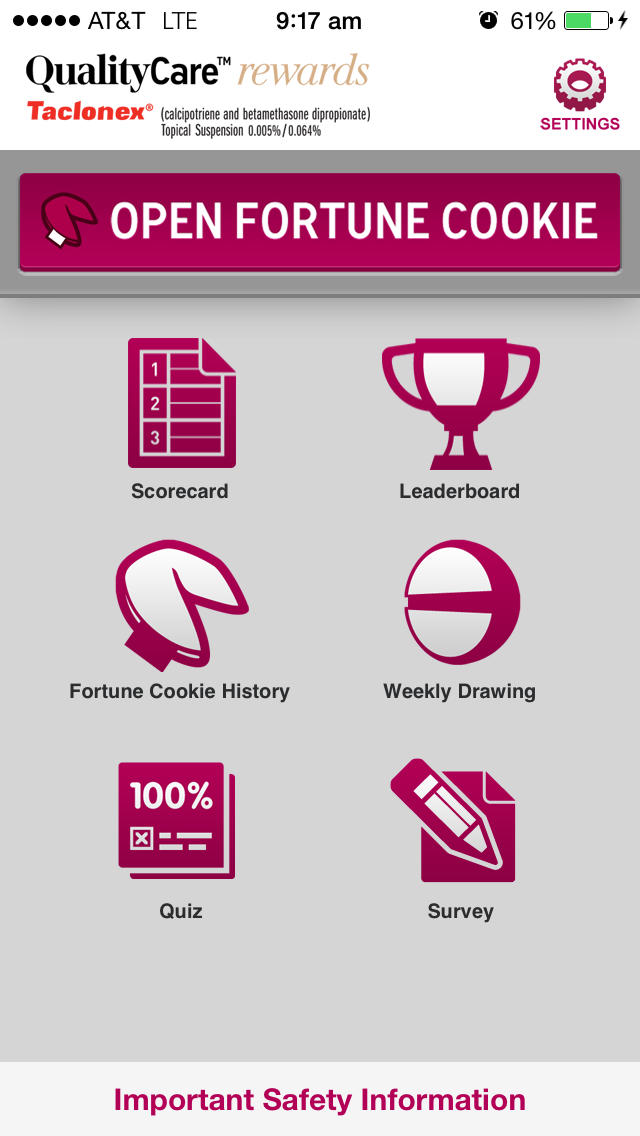
The free QualityCare™ rewards app is the mobile companion to the website version. Learning about psoriasis can be fun and rewarding. Earn points, prizes, and more on the go!
The mobile app offers these features:
•Open the daily Fortune Cookie
•Check your Scorecard
•See Weekly Drawing results
•Check your Leaderboard status
•Complete the weekly Survey
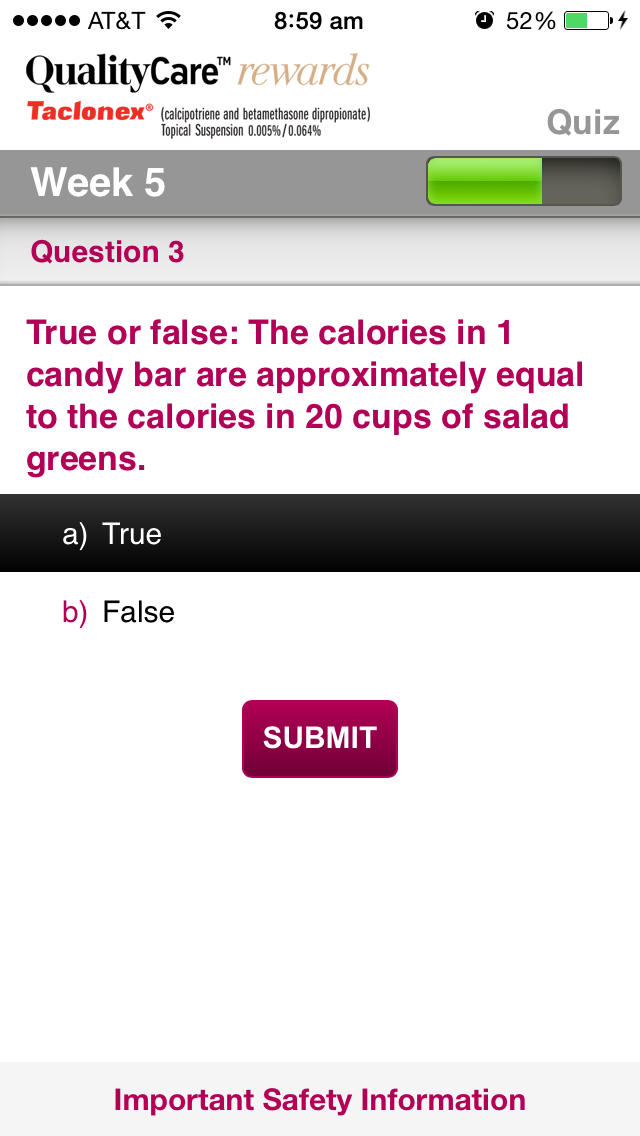
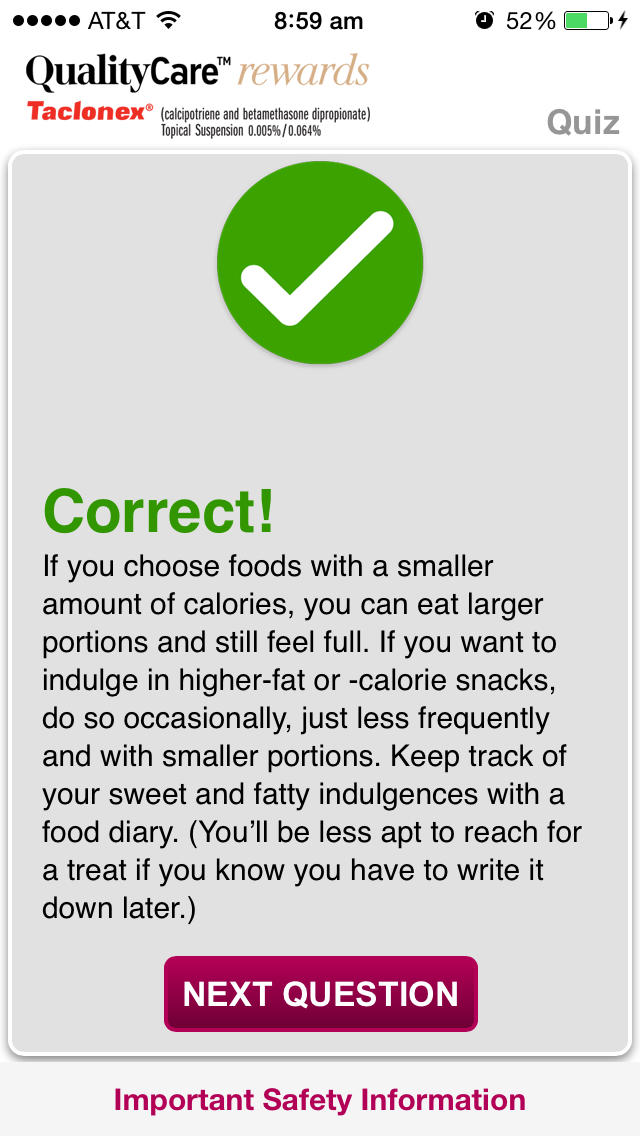
•Complete the weekly Quiz
•Change your settings
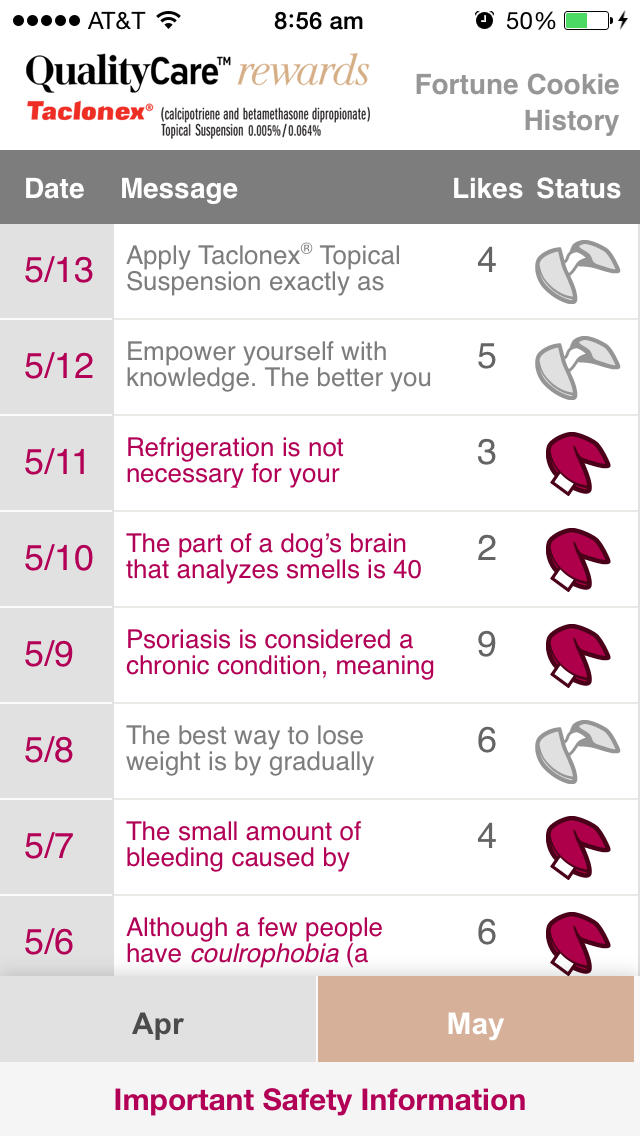
•View Fortune Cookie History
These features are “live” for registered users of QualityCare™ rewards online program only. Visit qualitycarerewards.com for more information on the program and visit Taclonex.com to learn more about Taclonex® (calcipotriene and betamethasone dipropionate) Topical Suspension and to obtain the Full Prescribing Information.
Indication: Taclonex® Topical Suspension is approved to be used on the skin to treat plaque psoriasis on the scalp and body in people 18 years and older. Apply Taclonex® Topical Suspension to areas affected by psoriasis 1 time a day for up to 8 weeks. Treatment may be discontinued earlier if cleared. Do not use more than the maximum recommended weekly amount of 100 grams. Use Taclonex® Topical Suspension exactly as your doctor tells you to use it.
Important Safety Information: Taclonex® Topical Suspension is for use on skin only (topical). Do not get Taclonex® Topical Suspension near or in your mouth, eyes, or vagina.
Taclonex® Topical Suspension may cause serious side effects including too much calcium in your blood or urine and adrenal gland problems. You may experience skin problems such as thinning of your skin, burning, inflammation, itching, irritation, dryness, changes in skin color, redness, infection, or raised bumps on your skin. You may get eye irritation if you accidently get Taclonex® Topical Suspension in your eyes. The most common side effects in the clinical trials were inflamed pores (folliculitis) and skin burning.
Tell your doctor if you have a skin infection, have a calcium metabolism disorder, have erythrodermic, exfoliative, or pustular psoriasis, have thinning-skin (atrophy) at the site to be treated, are getting light therapy (phototherapy treatments) for your psoriasis, are pregnant or plan to become pregnant, or are breastfeeding or plan to breastfeed. Tell your doctor about all the medicines you take, especially other corticosteroid medicines and other medicines for your psoriasis. Avoid spending a long time in sunlight. Avoid tanning booths and sun lamps.
If you have any questions or comments related to your health and wellness, medication, or other forms of treatment, please contact your healthcare provider. You are also encouraged to report negative side effects of prescription drugs to the FDA at www.fda.gov/medwatch or 1-800-FDA-1088, or LEO Pharama Inc. at 1-877-494-4536, and select option 1.
**IMPORTANT NOTICE** Apple is not a Sponsor of any of the sweepstakes or competitions conducted by LEO Pharma or HealthPrize as part of the QualityCare™ rewards program, and is not involved with LEO Pharma or HealthPrize, or any of the sweepstakes or competitions they conduct. See Sweepstakes at www.healthprize.com for more details.
less