Annual subscription with compliments from Roche Laboratory for doctors specialized in Oncology to Vademecum Internacional in the version for BlackBerry®, with all the information of Vademecum.es, the most visited Spanish pharmacologic website.
IF YOU ARE A DOCTOR SPECIALIZED IN ONCOLOGY AND YOU ARE INTERESTED IN DOWNLOADING THE APPLICATION, PLEASE CONTACT YOUR ROCHE REPRESENTATIVE TO OBTAIN AN ACTIVATION CODE.
*To download the application, introduce your Vademecum user information (if you are not a user yet, register free in www.vademecum.es), the activation code of the card and then access to the application. For more information, please contact Roche Farma S.A.
FUNCTIONALITIES
Information by active ingredients:
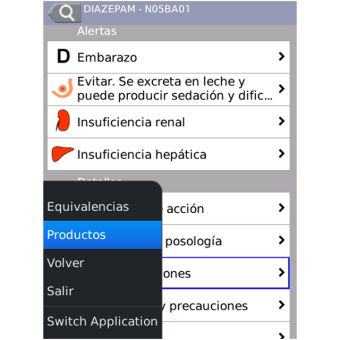
- Pharmacovigilance alerts, breastfeeding, pregnancy, kidney and liver failure, photosensitivity, effects on driving capability.
- Detailed monograph: dosage, therapeutic indications, warnings and precautions, contraindications, adverse reactions, interactions and a lot more.
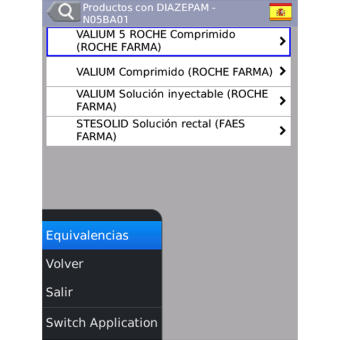
- Brand names of drugs in 28 countries, and sales information of Spanish drugs.
- Mabthera®, Avastin®, Herceptin®, Tarceva®y Xeloda® updated monographs
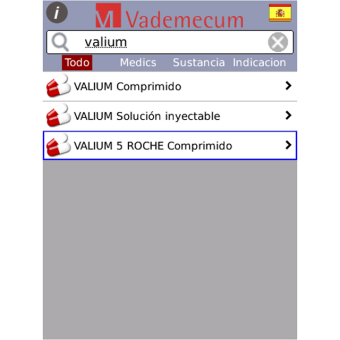
Search by:
-Indication.
- Active ingredient.
- Drug names in 28 countries.
Free regular updates.
AVAILABLE INFORMATION
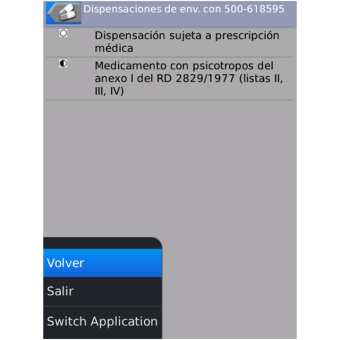
Active ingredient information for all commercialized drugs within the Spanish market: packages, pricing, national codes and dispensing information.
Information regarding active ingredients from over 5.000 brand names in México, Germany, France, United States, Italy, Portugal, Switzerland, Luxembourg and Chile (no information about pricing, national codes, etc. for drugs of these countries).
Information regarding active ingredients from up to 5.000 drugs in Sweden, Greece, Poland, Ireland, Austria, United Arab Emirates, Belgium, Netherlands, Hungary, Denmark, Morocco, Australia, Finland, Tunisia, Norway, Japan and Algeria (no information about pricing, national codes, etc. for drugs of these countries).
This application is addressed to healthcare professionals, and never has to be used as a substitute of a doctor's visit. The clinical decisions are an exclusive responsibility of the doctor in charge of the treatment. The Publishing Company verified carefully the data and its transcription. Nevertheless there is always a chance of unnoticed mistakes.
SYSTEM REQUIREMENTS AND TERMS OF USE
- You need to have 92Mb available space in the memory of your device, the required device software is 5.0.0 or higher. –Internet access needed for software activation. – Wi-Fi connection on the BlackBerry® required for updates. – Normal functionality does not require internet connection.
- Have a Vademecum user account (free registration).
- The license allows the installation of the application in two different devices maximum.
The Vademecum Roche Oncology application is free of charge for collaborators of Roche Laboratory.
less